Tianeptine
Dr. Michael P. Ludwig
© Dr Michael P. Ludwig 2014
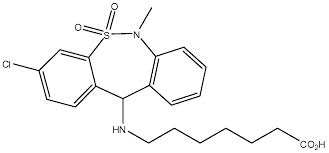
7-[(3-chloro-6-methyl-5,5-dioxo-11H-benzo[c]
[2,1]benzothiazepine-11-yl)amino]heptanoic acid
Tianeptine free acid
[2,1]benzothiazepine-11-yl)amino]heptanoic acid
Tianeptine free acid
Disclaimer: Nothing presented in this article is to be construed as constituting
**** medical advice or pharmaceutical consultation. This article is intended purely ****
as general knowledge and entertainment
**** medical advice or pharmaceutical consultation. This article is intended purely ****
as general knowledge and entertainment
Conflict of Interest and Funding Disclosure: Dr. Michael P. Ludwig possesses no conflicts of interests of any kind.
INTRODUCTION
Tianeptine is an atypical antidepressant with a novel mechanism of action which shows efficacy in depressed patients who are refractory to standard antidepressant treatment⁽³⁰⁾. When used in recommended dosages, it exhibits a favorable adverse reaction, or side effect profile when compared to standard antidepressants⁽²⁴⁾. Improvement in mood is seen nearly immediately after beginning therapy, whereas most antidepressants take weeks to show improvement and can even initially worsen symptoms of low mood and depression. As well, emotional blunting, which can be seen in many typical antidepressants, is not observed with the use of tianeptine. Despite all this, and the fact that tianeptine is available as a prescription drug throughout Europe, Asia and South America, it has never been approved for medicinal use in North America, leaving patients residing in the United States and Canada without prescription access to this compound.
It is possible that some readers in the United States or Canada, have yet to learn of tianeptine. Like some of the drugs in the racetam class, tianeptine has been approved for human use and sold as a prescription medication in many other countries. There is a good chance that if one were to travel to France and consult a pharmacist about Stablon®, they would know exactly what was meant, as Stablon® is the brand name for tianeptine in France. In Russia, tiapeptine is known as Coaxil® which would also be easily recognizable to any competent pharmacist. Despite being utilized as a successful treatment modality in Europe, Asia and South American, tianeptine has never been approved for human use in the United States nor Canada.
Where as the racetam class of drugs are usually thought of for their memory enhancing effects, tianeptine is usually thought first and foremost as an antidepressant, because, well, that is what it is. In France, it is officially approved only for major depressive disorder⁽²⁴⁾, while in Malaysia it is approved for neurotic and reactive depression, anxiodepressive states with psychosomatic manifestations, especially gastrointestinal, and anxiodepressive states in alcoholics during withdrawal stage⁽²⁵⁾. These indications are based on the two complete package inserts this author could locate. It is possible that other countries may have different indications.
MECHANISM OF ACTION
The exact mechanism of action(MOA) of tianeptine is still not completely understood. For a time, the sole mechanism was attributed to selective serotonin re-uptake enhancement(SSRE )⁽¹⁶⁾, which despite sounding counter-intuitive, would be a viable mechanism for anti depression. It is now commonly agreed upon that the antidepressant effects of SSRI's are partly due to down-regulation of 5-HT₂ₐ receptors⁽⁴⁾, which is a plausible explanation for the long onset of action of antidepressant effects, as it takes some time for the receptors to down-regulate to an appreciable level. Selective re-uptake enhancement would provide less activation of 5-HT₂ₐ receptors without having to wait for the receptors to down-regulate. Tianeptine's effect on serotonin in the prefrontal cortex has been disputed, though. Some research has shown the original study that suggested the SSRE MOA to have been flawed. Furthermore, newer research has shown tianeptine to have no effect on serotonin levels in the prefrontal cortex⁽¹⁴⁾.
Glutamate is the main excititory neurotransmitter in the brain and acts on a variety of receptor types. Tianeptine has been shown to modulate glutamate activity in the hippocampus at the ionotropic receptors: NMDA, AMPA, and Kainate⁽¹¹· ²⁶· ³²⁾. Chronic stress has been shown to cause disregulation in hippocampal glutamate signaling, specifically increasing the ration of NMDA to AMPA and Kainate signaling. Tianeptine, in the rat model, has been shown to normalize this ratio⁽¹¹⁾. Tianeptine exhibits no affinity for any of the ionotropic glutamate receptors. It appears the regulatory effects of tianeptine on glutamate are secondary to some other mechanism.
Tianeptine has been shown to increase dopamine levels in the nucleus accumbens (NAc)⁽⁹⁾. The NAc along with its projections from the ventral tegmental area (VTA) and projections to the thalamus and prefrontal cortex (PFC) plays a major role in reinforcement and reward. Decreased basal levels of dopamine could be a possible cause of anhedonia, therefore the ability of tianeptine to indirectly increase dopamine levels in the NAc could be associated with decreased anhedonia.
INTRODUCTION
Tianeptine is an atypical antidepressant with a novel mechanism of action which shows efficacy in depressed patients who are refractory to standard antidepressant treatment⁽³⁰⁾. When used in recommended dosages, it exhibits a favorable adverse reaction, or side effect profile when compared to standard antidepressants⁽²⁴⁾. Improvement in mood is seen nearly immediately after beginning therapy, whereas most antidepressants take weeks to show improvement and can even initially worsen symptoms of low mood and depression. As well, emotional blunting, which can be seen in many typical antidepressants, is not observed with the use of tianeptine. Despite all this, and the fact that tianeptine is available as a prescription drug throughout Europe, Asia and South America, it has never been approved for medicinal use in North America, leaving patients residing in the United States and Canada without prescription access to this compound.
It is possible that some readers in the United States or Canada, have yet to learn of tianeptine. Like some of the drugs in the racetam class, tianeptine has been approved for human use and sold as a prescription medication in many other countries. There is a good chance that if one were to travel to France and consult a pharmacist about Stablon®, they would know exactly what was meant, as Stablon® is the brand name for tianeptine in France. In Russia, tiapeptine is known as Coaxil® which would also be easily recognizable to any competent pharmacist. Despite being utilized as a successful treatment modality in Europe, Asia and South American, tianeptine has never been approved for human use in the United States nor Canada.
Where as the racetam class of drugs are usually thought of for their memory enhancing effects, tianeptine is usually thought first and foremost as an antidepressant, because, well, that is what it is. In France, it is officially approved only for major depressive disorder⁽²⁴⁾, while in Malaysia it is approved for neurotic and reactive depression, anxiodepressive states with psychosomatic manifestations, especially gastrointestinal, and anxiodepressive states in alcoholics during withdrawal stage⁽²⁵⁾. These indications are based on the two complete package inserts this author could locate. It is possible that other countries may have different indications.
MECHANISM OF ACTION
The exact mechanism of action(MOA) of tianeptine is still not completely understood. For a time, the sole mechanism was attributed to selective serotonin re-uptake enhancement(SSRE )⁽¹⁶⁾, which despite sounding counter-intuitive, would be a viable mechanism for anti depression. It is now commonly agreed upon that the antidepressant effects of SSRI's are partly due to down-regulation of 5-HT₂ₐ receptors⁽⁴⁾, which is a plausible explanation for the long onset of action of antidepressant effects, as it takes some time for the receptors to down-regulate to an appreciable level. Selective re-uptake enhancement would provide less activation of 5-HT₂ₐ receptors without having to wait for the receptors to down-regulate. Tianeptine's effect on serotonin in the prefrontal cortex has been disputed, though. Some research has shown the original study that suggested the SSRE MOA to have been flawed. Furthermore, newer research has shown tianeptine to have no effect on serotonin levels in the prefrontal cortex⁽¹⁴⁾.
Glutamate is the main excititory neurotransmitter in the brain and acts on a variety of receptor types. Tianeptine has been shown to modulate glutamate activity in the hippocampus at the ionotropic receptors: NMDA, AMPA, and Kainate⁽¹¹· ²⁶· ³²⁾. Chronic stress has been shown to cause disregulation in hippocampal glutamate signaling, specifically increasing the ration of NMDA to AMPA and Kainate signaling. Tianeptine, in the rat model, has been shown to normalize this ratio⁽¹¹⁾. Tianeptine exhibits no affinity for any of the ionotropic glutamate receptors. It appears the regulatory effects of tianeptine on glutamate are secondary to some other mechanism.
Tianeptine has been shown to increase dopamine levels in the nucleus accumbens (NAc)⁽⁹⁾. The NAc along with its projections from the ventral tegmental area (VTA) and projections to the thalamus and prefrontal cortex (PFC) plays a major role in reinforcement and reward. Decreased basal levels of dopamine could be a possible cause of anhedonia, therefore the ability of tianeptine to indirectly increase dopamine levels in the NAc could be associated with decreased anhedonia.
Tianeptine has recently been show to have affinity for the mu opioid receptor (MOR) and the delta opioid receptor (DOR)⁽⁷⁾. To date, these are the only receptors that tianeptine has shown any appreciable affinity for. It is possible that the effects of tianeptine on the dopamine, serotonin and glutamate systems are all down stream effect of its MOR activity.
Tianeptine effects MOR to a much greater degree than the delta opioid receptor (DOR), and has little to no effect on the kappa opioid receptor (KOR). Of the major endogenous opioid peptides, the binding profile of tianeptine resembles that of the endorphins rather than the enkephalins or dynorphins.
Althgough tianeptine binds to and has agonist activity at MOR, its affinity is rather weak when compared to other exogenous MOR agonists. Despite its low affinity, tianeptine does have minor agonistic effect on MOR at therapeutic dosages of 12.5mg taken three times daily⁽⁷⁾. This minor agonistic effect can almost be thought of as a microdosing effect. In the model about to be proposed, tianeptine, at therapeutic dosages acts in a microdosing manner that helps regulate a feedback loop that exists between MOR and NMDAR while at the same time initiating a cascade that leads to the phosphorylation that ultimately regulates the NMDAR/AMPAR and Kainait receptors. Activation of MOR, in this model, would also be responsible for the increase in dopamine seen in the Nac. If this model is valid, the reader could possibly see that all the various effects of tianeptine begins at MOR with everything else being downstream results.
One can suggested, that through its low affinity for MOR, tianeptine, acting either in a microdosing manner, or in the manner of a yet to be discovered trace opioid peptide, balances a feedback loop that exists between some MOR and NMDARs.
Tianeptine activates MOR which is functionally connected to NMDAR via histidine triad nucleotide-binding protein 1(HINT1). This is essentially the backbone of a negative feedback loop that ultimately involves PKC, CaMKII, Src, and NOS1⁽¹⁸·¹⁹·²⁰·²²·²³⁾.
When MOR is activated via an agonist, the result is a reduction-oxidation(redox) reaction that causes zinc binding protein to release free zinc ions via a nitric oxide synthase 1(NOS1)/nitric oxide(NO) dependent mechanism⁽¹⁹⁾.
MOR is a G-protein coupled receptor(GPCR) as opposed to an ionotropic receptor. Ionotropic receptors, when activated, directly affect the opening and closing of ion gates therefore controlling the flow of ions such as calcium into the cell. GPCRs, when activated, do not cause their effect via an ion gate, instead they directly activate signaling cascades within the cell. This is possible, because part of the GPCR exist on the outside of the cell while part resides inside the cell. GPCR's are also known as seven-transmembrane receptors since they loop in and out of the cell seven times. This is important to know, because it is the GPCR that regulates the zinc ion release via the NOS1/NO mechanism. It is possible that any dysfunction of this pathway may possibly lead to depression.
Normally when MOR activation leads to the release of zinc ions via the NOS1/NO mechanism there is an accompanied release of nitric oxide (NO). NO directly activates calcium/calmodulin-dependent protein kinase II(CaMKII) even in the absence of calcium⁽²³⁾.
The activated CaMKII is now able to desensitize MOR. The zinc ions that were released along with the NO activate protein kinase c (PKC). Activated PKC now phosphorylates the terminal serine on the alpha subunit of the MOR G protein. This reduces the signal strength from MOR. Together with the CaMKII mediated desensitization of MOR, the result is a negative feedback loop designed to prevent over activation or MOR⁽⁶⁾.
How does this MOR-NMDAR feedback loop result in the downstream effects, specifically on the glutamate and the dopamine systems?
Phosphorylation of NMDARs plays a big role in the receptors behavior⁽¹·²⁾. Depending on which subunit is phosphorylated by which kinase determines how the NMDAR behaves. During the cascade in the MOR-NMDAR feedback loop there are multiple opportunities for the NMDAR to be phosphorylated. When CaMKII is activated by NO it could directly phosphorylate the NMDAR. Activated PKC can also phosphorylate NMDAR. It's likely that phosphorylation by one of these two enzymes, CaMKII or PKC, at either the NR1 or NR2 subunit of the NMDAR is responsible for increased activity of said NMDAR which plays a part in the increased ratio of NMDAR to AMPAR and Kainate activity.
Separately, the protein ΔfosB may also play a role in the disregulation of the ratio of NMDAR to AMPAR activity⁽²⁹·³⁰⁾. During periods of chronic stress, serum response factor(SRF) causes ΔfosB to accumulate in the NAc. Initially, the increased ΔfosB helps by providing resilience toward the stress, but when the stress remains chronic, the (SRF) begins to drop and so does the ΔfosB. As the levels of ΔfosB fall, so does the subject's resilience and ability to handle stress⁽²⁹·³⁰⁾.
ΔfosB is a transcription factor that seems to play a role in either up-regulation or down-regulation of GluRA2 subunits. The relationship seems to be direct, such that increased ΔfosB leads to increased expression of GluRA2 subunits.
GluRA2 subunits are the AMPA receptor subunits that help prevent exitotoxicity by limiting the AMPAR channel to only allow influx of potassium and not calcium.
The action of tianeptine on MOR possibly leads to increased levels of ΔfosB which in turn could lead to increased expression of GluR2A subunits. Increased GluR2A subunits are associated with increased resilience to stress⁽²⁹·³⁰⁾, therefore tianeptine's action at MOR can bring the ΔfosB levels in the NAc back to levels associated with resilience and the ability to handle stress in a non depressive manner.
Activation of MOR causes the VTA to project dopamine to the NAc, the PFC, and the hippocampus. The mesolimbic and mesocortical dopamine pathways and their relationship to MOR is well established.
In the end, tianeptine ultimately effects many neuronal systems including serotonin, dopamine, glutamate, endogenous opioids, and possible many other systems. No one, at this time, can point to a definitive mechanism of action, though the proof of tianeptine's affinity for MOR is a step in the right direction. This author believes that the therapeutic mechanism of action of tianeptine is in its the ability to regulate a disregulation in the MOR-NMDAR feedback loop.
ABUSE POTENTIAL
Case reports of tianeptine abuse and dependence go as far back as 1999⁽²⁸⁾; fifteen years before its opioid activity was discovered. At its recommended dose of 12.5mg, it is therapeutic with a favorable side effect profile. At supra-therapeutic doses the subjective effect of the drug becomes recreational and thus exhibits potential for abuse. There have been cases of patients “doctor shopping” in France, in attempt to obtain prescription tianeptine⁽²¹⁾. Doctor shopping is the practice of visiting multiple physicians with the intent of obtaining multiple prescriptions for a desired drug. Doctor shopping happens in the United States as well, but the desired drug is usually a much more powerful narcotic. Oxycodone, oxymorphone, and hydromorphone are more likely to be doctor shopped in the United States . Those three drugs are some of the most powerful, abuseable narcotics in use, yet people in France attempt to obtain tianeptine in the same manner that people in the United States attempt to obtain them.
There have been cases of people injecting tianeptine, not only intravenously, but intraarterially. In a Turkish case report, a patient with a large swollen area on his right leg near his groin. The mass was the external view of what was determined to be a pseudoaneurysm, which was the result of intraarterial injection of disolved tianeptine pills. The vessel was successfully repaired and the patient sent home. Two months later he returned to the hospital with the same presentation, only on his other leg⁽⁸⁾.
There is no doubt that tianeptine can exhibit abuse potential. Much of the abuse has happened abroad⁽¹²·²⁷⁾. Now that it is widely known that tianeptine is a low potency MOR agonist and that supra-therapeutic doses are able to negate this low potency, One must wonder how long it will take before tianeptine becomes one of the DEA's “drugs of concern.”
This author just hopes that enough people are responsible with this drug, so as outshine the few who choose to abuse it. This is one that this author really hopes does not get ruined by the DEA. The abuse potential of tianeptine is undeniable, however the benefits of the drug far outweigh the risks.
PHYSICAL PROPERTIES
Tianeptine sodium is a rather hygroscopic compound. This means it has a tendency to absorb water from the air. This causes two main problems. At first, it makes the powder hard to deal with, as it will begin to stick and clump to just about anything. The second main problem is that after the tianeptine has absorbed any appreciable amount of water, your weights will be off; when weighing the powder, you are now also weighing any water is it has absorbed.
Fortunately, the same love for water that makes tianeptine hygroscopic also allows for it to easily dissolve into solution.
Be sure to view the step-by-step tutorial on how to compound an H₂O or PG solution with tianeptine sodium powder. •••• Disclaimer: the step-by-step tutorial is intended only as an educational device concerning dissolution of a single solute into multiple solvents. The product of the tutorial is not intended for human consumption, as tianeptine, despite being proven safe and effective throughout the world, is a non FDA approved medication ••••
If any readers lack the required equipment confidence to make their own solution, I personally recommend the solution available at www.ceretropic.com
DOSING
The recommend dosage is 12.5mg taken three times daily with meals⁽²⁴⁾. This author would not recommend pushing the dosage too much higher. There is a point at which this compound can go from therapeutic to recreational. The package insert recommends that patients with renal insufficiency cut the dosage down to 12.5mg twice daily. The package insert did not specify a particular degree of renal insufficiency, though, to be safe, this author would like to point out that anyone with a glomerular filtration rate less than 100 can be potentially considered renal insufficient. The package insert also stated that anyone over the age of 70 should cut the dosage down to 12.5mg twice daily, though there was not mention of why this is necessary. If discontinuing tianeptine therapy, one should gradually taper their dosage over the course or one to two weeks⁽²⁴⁾.
DRUG INTERACTIONS
Monoamine oxidase inhibitors (MAOIs) are contraindicated with the use of tianeptine. There needs to be at least a 14 day washout period after stopping MAOI therapy and beginning tianeptine therapy. Conversely, there needs to be a 24 hour washout period after stopping tianeptine therapy and beginning MOAI therapy⁽²⁴⁾. MAO-B selective inhibitors such as selegeline and rasagiline, when taken at lower dosages, may potentially be a relative contraindication with tianeptine. Relative contraindications, as opposed to absolute contraindications, may allow, in certain conditions, for the drugs to be taken together, though this is a combination one should consult their physician about. It is important to note that the MAO-B selective inhibitors both lose their selectivity as dosages increase, therefore high doses of selegeline and rasagiline would both be considered absolute contraindications.
Tianeptine is also contraindicated with the antidepressant, mianserin⁽²⁴⁾. Mianserin is not used in the United States, though mirtazapine, which is a close structural analogue to mianserin is routinely used in the United States, therefore there may potentially exist a drug-drug interaction between tianeptine and mirtazepine.
PREGNANCY AND BREASTFEEDING
Use of tianeptine is generally to be avoided during pregnancy, though Servier's package insert states “Animal trials are reassuring, but clinical data is still insufficient.” This translates roughly into what would be considered a Pregnancy Category B drug in the United States. Usage of drugs in this category ultimately come down to a decision made between the expecting parent(s) and the physician after all possible risks and benefits of continuing the drug through pregnancy are considered. Many category B drugs are routinely used during pregnancy. There are fairly few category A drugs; category A being drugs that are considered completely safe during pregnancy.
Tianeptine is not recommended during breastfeeding⁽²⁴⁾,
POSSIBLE SIDE EFFECTS
Tianeptine, at its therapeutic dosage seems to be pretty well tolerated. The most commonly reported side effect seems to be sleepiness. Servier's package insert for Stablon® includes a list of all reported side effects from their trials, which is inserted below:
“As with any active pharmaceutical product, this medicine may cause somewhat unpleasant side effects in certain patients. Although such effects are rare, usually mild and temporary, they include: abdominal pain, dryness of the mouth, anorexia, nausea, vomiting, flatulence, insomnia, drowsiness, nightmares, asthenia, tachycardia, extrasystole, precordialgia, dizziness, headaches, faintness, trembling, respiratory discomfort, tightness of the throat, myalgia, lumbago, abuse and/or dependence especially in patients under 50 years of age with a history of drug or alcohol dependence. Cases of suicidal thoughts or behavior have been reported during treatment or shortly after its discontinuation.”
Most of the side effects listed above are rare and do not occur in most individuals. Some of the adverse reactions listed are common to many drugs, however others are worth pointing out. Asthenia, which translates to a general weakness could be a result of the sleepiness or somnolence caused by the drug, though any severe weakness especially with rapid onset would be cause for concern. Extrasystole and tachycardia, meaning an extra heartbeat and a fast heartbeat(>100 beats per minute) respectively, could range from generally benign to quite severe. Precordalgia, which is a pain felt somewhere under the upper rib cage, usually on the left side. The pain can be quite severe though it is usually benign. Some readers may have experienced precordialgia in the form of a precordial catch when they were younger. The pain, usually described as sharp and stabbing, would typically occur during deep breathes either in or out and would resolve after a few minutes. Respiratory discomfort and tightness of the throat are both causes for concern and could be signs of an allergic reaction. Myalgia, or muscle pain, could be cause for concern. Myalgia could be a sign of rhabdomyolysis, or muscle breakdown. The resulting proteins from broken down muscle can cause potentially serious kidney damage including kidney failure. Lumbago is a general lower back pain and is a very nondescript symptom. Suicidal thoughts, ideation, or behavior are always to be taken seriously.
GENERAL ANESTHESIA
If one is to be put under general anesthesia they must inform their anesthesiologist of their tianeptine usage and discontinue usage for one to two days prior to surgery⁽²⁴⁾. It is normally the responsibility of the anesthesiologist to inform patients which medications they should discontinue, however, with tianeptine not being an approved drug in the United States, this author advices that patients take it upon themselves to inform their anesthesiologist of their tianeptine use, or use of any other unapproved drugs. Drugs such as tianeptine, piracetam, phenibut, etc... are not routinely taught in American medical schools or American pharmacy schools.
CONCLUSION
Tianeptine is an interesting drug with a unique, if not 100% understood, mechanism of action. It is the only antidepressant known to work in the manner in which it does. It may get structurally grouped in with the tricyclic antidepressants, but functionally it is truly in a class by itself. It works fast, it has a favorable side effect profile, and it helps people who have failed to respond to other antidepressants. This drug should be available, by prescription, to American and Canadian citizens. Even with the minor abuse potential, the benefits outweigh the risks. If abuse were a concern, the drug could get scheduled into CV or CIV.
AUTHOR AVAILABLE FOR CORRESPONDENCE
Dr. Ludwig would be happy to answer any questions that one may have.
Please do not hesitate to contact. He can be reached at [email protected]
Questions about the article as well as general questions are welcome. Dr. Ludwig is always open to discussion regarding the underlying mechanisms of addiction and depression. He takes special interest in the transcription factors involved, especially ΔfosB. Experimental treatments for depressionn, including those involving ketamine and its analogs, and serotonin releasing experimental treatments involving novel MDMA analogues. Dr. Ludwig is a big fan of Dr. David Nichols and the work he has done at Purdue and elsewhere.
WORKS CITED
1. Chen BS, Braud S, Badger JD 2nd, Isaac JT, & Roche KW. (2006). Regulation of NR1/NR2C N-methyl- D-aspartate (NMDA) receptors by phosphorylation. The Journal of Biological Chemistry. 281(24), 16583-90.
2. Chen BS, & Roche KW. (2007). Regulation of NMDA receptors by phosphorylation. Neuropharmacology. 53(3), 362-8.
3. Della FP, Abelaira HM, Reus GZ, Ribeiro KF, Antunes AR, Scaini G, Jeremias IC, dos Santos LM, Jeremias GC, Streck EL, & Quevedo J. (2012). Tianeptine treatment induces antidepressive-like effects and alters BDNF and energy metabolism in the brain of rats. Behavioural Brain Research. 233(2), 526-35.
4. Eison AS, & Mullins UL. (1996). Regulation of central 5-HT2A receptors: a review of in vivo studies. Behavioural Brain Research. 73(1-2), 177-81.
5.Garzon J, Rodriguez-Munoz M, & Sanchez-Blazquez P. (2012). Direct association of Mu-opioid and NMDA glutamate receptors supports their cross-regulation: molecular implications for opioid tolerance. Current Drug Abuse Reviews. 5(3), 199-226.
6. Garzon J, Rodriguez-Munoz M, Vicente-Sanchez A, Bailon C, Martinez-Murillo R, & Sanchez- Blazquez P. (2011). RGSZ2 binds to the neural nitric oxide synthase PDZ domain to regulate mu- opioid receptor-mediated potentiation of the N-methyl-D-aspartate receptor-calmodulin- dependent protein kinase II pathway. Antioxidants & Redox Signaling. 15(4), 873-87.
7. Gassaway MM, Rives ML, Kruegel AC, Javitch JA, & Sames D. (2014). The atypical antidepressant and neurorestorative agent tianeptine is a o'-opioid receptor agonist. Translational Psychiatry. 4, e411.
8. Ilhan G, Ergene S, Durakoglugil T, Karamustafa H, Karakis'i O, & Bozok S. (2013). Bilateral pseudoaneurysm secondary to intraarterial tianeptine abuse. Anadolu Kardiyoloji Dergisi : AKD = the Anatolian Journal of Cardiology. 13(8), 814-5.
9. Invernizzi R, Pozzi L, Garattini S, & Samanin R. (1992). Tianeptine increases the extracellular concentrations of dopamine in the nucleus accumbens by a serotonin-independent mechanism. Neuropharmacology. 31(3), 221-7.
10. Jeon HJ, Woo JM, Lee SH, Kim EJ, Chung S, Ha JH, Fava M, Mischoulon D, Kim JH, Heo JY, & Yu BH. (2014). Improvement in subjective and objective neurocognitive functions in patients with major depressive disorder: a 12-week, multicenter, randomized trial of tianeptine versus escitalopram, the CAMPION study. Journal of Clinical Psychopharmacology. 34(2), 218-25.
11. Kole MH, Swan L, & Fuchs E. (2002). The antidepressant tianeptine persistently modulates glutamate receptor currents of the hippocampal CA3 commissural associational synapse in chronically stressed rats. The European Journal of Neuroscience. 16(5), 807-16.
12. Leterme L, Singlan YS, Auclair V, Le Boisselier R, & Frimas V. (2003). Usage detourne de tianeptine. Apropos de cinq ans de surconsommation. Annales De Medecine Interne. 154, S58-63.
13. McEwen BS, & Chattarji S. (2004). Molecular mechanisms of neuroplasticity and pharmacological implications: the example of tianeptine. European Neuropsychopharmacology : the Journal of the European College of Neuropsychopharmacology. 14, S497-502.
14. McEwen BS, Chattarji S, Diamond DM, Jay TM, Reagan LP, Svenningsson P, & Fuchs E. (2010). The neurobiological properties of tianeptine (Stablon): from monoamine hypothesis to glutamatergic modulation. Molecular Psychiatry. 15(3), 237-49.
15. McEwen BS, & Olie JP. (2005). Neurobiology of mood, anxiety, and emotions as revealed by studies of a unique antidepressant: tianeptine. Molecular Psychiatry. 10(6), 525-37.
16. Mennini T, Mocaer E, & Garattini S. (1987). Tianeptine, a selective enhancer of serotonin uptake in rat brain. Naunyn-Schmiedeberg's Archives of Pharmacology. 336(5), 478-82.
17. Qi H, Mailliet F, Spedding M, Rocher C, Zhang X, Delagrange P, McEwen B, Jay TM, & Svenningsson P. (2009). Antidepressants reverse the attenuation of the neurotrophic MEK/MAPK cascade in frontal cortex by elevated platform stress; reversal of effects on LTP is associated with GluA1 phosphorylation. Neuropharmacology. 56(1), 37-46.
18. Rodriguez-Munoz M, de la Torre-Madrid E, Sanchez-Blazquez P, & Garzon J. (2011). NO-released zinc supports the simultaneous binding of Raf-1 and PKCd cysteine-rich domains to HINT1 protein at the mu-opioid receptor. Antioxidants & Redox Signaling. 14(12), 2413-25.
19. Rodriguez-Munoz M, & Garzon J. (2013). Nitric oxide and zinc-mediated protein assemblies involved in mu opioid receptor signaling. Molecular Neurobiology. 48(3), 769-82
20. Rodriguez-Munoz M, Sanchez-Blazquez P, Vicente-Sanchez A, Bailon C, Martin-Aznar B, & Garzon J. (2011). The histidine triad nucleotide-binding protein 1 supports mu-opioid receptor- glutamate NMDA receptor cross- regulation. Cellular and Molecular Life Sciences : CMLS. 68(17), 2933-49.
21. Rouby F, Pradel V, Frauger E, Pauly V, Natali F, Reggio P, Thirion X, & Micallef J. (2012). Assessment of abuse of tianeptine from a reimbursement database using 'doctor-shopping' as an indicator. Fundamental & Clinical Pharmacology. 26(2), 286-94.
22. Sanchez-Blazquez P, Rodriguez-Munoz M, Berrocoso E, & Garzon J. (2013). The plasticity of the association between mu-opioid receptor and glutamate ionotropic receptor N in opioid analgesic tolerance and neuropathic pain. European Journal of Pharmacology. 716(1-3), 94- 105.
23. Sanchez-Blazquez P, Rodriguez-Munoz M, & Garzon J. (2010). Mu-opioid receptors transiently activate the Akt-nNOS pathway to produce sustained potentiation of PKC-mediated NMDAR- CaMKII signaling. PloS One. 5(6), e11278.
24. Stablon® Package Insert, French
25. Stablon® Package Insert, Malaysian
26. Szegedi V, Juhasz G, Zhang X, Barkoczi B, Qi H, Madeira A, Kapus G, Svenningsson P, Spedding M, & Penke B. (2011). Tianeptine potentiates AMPA receptors by activating CaMKII and PKA via the p38, p42/44 MAPK and JNK pathways. Neurochemistry International. 59(8), 1109-22.
27. Vadachkoria D, Gabunia L, Gambashidze K, Pkhaladze N, & Kuridze N. (2009). Addictive potential of Tianeptine - the threatening reality. Georgian Medical News. (174), 92-4.
28. Vandel P, Regina W, Bonin B, Sechter D, & Bizouard P. (1999). Abus de tianeptine. A propos d'un cas. L'Encephale. 25(6), 672-3.
29. Vialou V, Maze I, Renthal W, LaPlant QC, Watts EL, Mouzon E, Ghose S, Tamminga CA, & Nestler EJ. (2010). Serum response factor promotes resilience to chronic social stress through the induction of DeltaFosB. The Journal of Neuroscience : the Official Journal of the Society for Neuroscience. 30(43), 14585-92.
30. Vialou V, Robison AJ, Laplant QC, Covington HE 3rd, Dietz DM, Ohnishi YN, Mouzon E, Rush AJ 3rd, Watts EL, Wallace DL, Iniguez SD, Ohnishi YH, Steiner MA, Warren BL, Krishnan V, Bolanos CA, Neve RL, Ghose S, Berton O, Tamminga CA, & Nestler EJ. (2010). DeltaFosB in brain reward circuits mediates resilience to stress and antidepressant responses. Nature Neuroscience. 13(6), 745-52.
31. Woo YS, Bahk WM, Jeong JH, Lee SH, Sung HM, Pae CU, Koo BH, & Kim W. (2013). Tianeptine combination for partial or non-response to selective serotonin re-uptake inhibitor monotherapy. Psychiatry and Clinical Neurosciences. 67(4), 219-27.
32. Zhang H, Etherington LA, Hafner AS, Belelli D, Coussen F, Delagrange P, Chaouloff F, Spedding M, Lambert JJ, Choquet D, & Groc L. (2013). Regulation of AMPA receptor surface trafficking and synaptic plasticity by a cognitive enhancer and antidepressant molecule. Molecular Psychiatry. 18(4), 471-84.

 RSS Feed
RSS Feed
